Sugarcane (Saccharum spp.) is an important cash crop that contributes 80% of global consumption and 40-50% of the world’s bio-ethanol production (Zuurbier & Vooren, 2008). Nitrogen (N) is involved in many critical processes in plant growth, expansion of green leaves, and tiller production, especially in the formation of plant protein which is essential for photosynthesis. Previous studies showed that higher leaf N content resulting from higher N application supports better photosynthesis in sugarcane (Sage et al., 2014). The N requirement of sugarcane is the greatest during tillering and the early grand growth phases (Bachchhav, 2005). Application of more N promotes late tiller formation, reduces sugar recovery, increases N in juice, and attracts pests and diseases (Castro et al., 2017). Therefore, in sugarcane production, N is often supplied during the early growth stage from planting to elongating.
Water shortage frequently occurs during the early growth stage of sugarcane because of crop season management (Dinh et al., 2017). Fertilizer application with high doses of N during this stage supports stronger photosynthesis and also induces more transpiration. This agricultural practice implicit more risks from water stress if the water supply is limited. Hence, applying N efficiently to support sugarcane growth and using water sources sparingly are important strategies in sugarcane cultivation. Previous studies mostly investigated the effects of N on photosynthesis and the growth of sugarcane, whereas little information on transpiration, especially daily whole-plant transpiration, has been found. Therefore, a study was carried out at the University of the Ryukyus, Okinawa, Japan to study daily transpiration, growth, and physiological responses to different N applications. The results of the study would provide useful information to enhance N use efficiency in sugarcane at the early growth stage.
The pot experiment was conducted under glass-house conditions. Five treatments of various nitrogen application rates (0, 4, 8, 12, and 16 mM) were assigned in a randomized complete block design with three replications. Experimental pots were arranged between the plants and rows at 40cm and 90cm, respectively.
The experimental pots were weighed daily by an electronic digital scale at 6pm. The pot weight before and after irrigation was recorded. From 3 weeks after transplanting (WAT), growth parameters were weekly determined. Plant height was measured from ground to top visible dewlap leaf. Leaves were marked to count the number of green leaves and the total number of leaves from the beginning to the measured times. The soil plant analysis development (SPAD) index was measured at the first fully expanded leaf by a SPAD 502 Plus Chlorophyll Meter (Konica Minolta, Japan). Following that, the first leaf was cut to determine the leaf area using LI-3100 portable leaf area meter (LI-COR, Lincoln, Nebraska, USA).
At the end of the experiment (12 WAT), photosynthesis parameters, including potential photosynthesis rate, stomatal conductance, and transpiration rate, were measured at the first fully expanded leaf using LI-6400 portable photosynthesis system (LI-COR, Lincoln, Nebraska, USA) equipped with a 2x3cm2 LED chamber between 1100 to 1300 at a photon flux density of 2,000 μmol m-2 s-1, leaf temperature of 33 ± 2°C, the CO2 concentration of 400 ± 5 μmol mol-1.
    |
 |
| Measurement of photosynthesis using LI-6400 XT portable photosynthesis system |
N application supported better growth and physiological parameters of sugarcane. Higher N applications (12mM and 16mM) resulted in higher plant height, leaf number, SPAD, N and chlorophyll contents, potential photosynthesis, whole plant transpiration, and biomass than those treatments with lower N applications (0mM, 4mM, and 8mM). Nevertheless, biomass nitrogen use efficiency and photosynthetic water use efficiency declined because of applying nitrogen at higher rates.
    |
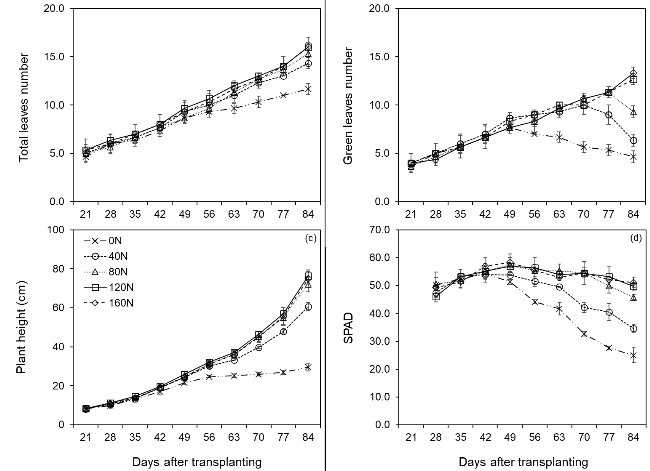 |
| Effects of nitrogen application on growth parameters of sugarcane |
    |
 |
| Effects of nitrogen on the growth of sugarcane |
During the first weeks of plant growth, no significant differences in plant height, leaf number, SPAD, and whole-plant transpiration among nitrogen treatments indicated that higher nitrogen added was not necessary during this time. Nitrogen and water use efficiencies could be improved if the remaining soil nitrogen and crop growth stage nitrogen requirements are estimated in cultivation practice.
The full paper can be accessed here on Vietnam Journal of Agricultural Sciences (ISSN 2588-1299).
References
Bachchhav M. (2005). Fertigation technology for increasing sugarcane production. Indian Journal of Fertilisers. 1(4): 85-92.
Castro S. G. Q. D, Neto J. R., Kölln O. T., Borges B. M. M. N. & Franco J. C. J. (2017). Decision-making on the optimum timing for nitrogen fertilization on sugarcane ratoon. Scientia Agricola. 76(3): 237-242.
Sage R. F., Peixoto M. M. & Sage T. L. (2014). Photosynthesis in sugarcane. In: Moore P.H. & Botha F. C. (Eds.). Physiology, biochemistry, and functional biology. Hoboken, NJ: Wiley: 377-410.
Zuurbier P. & Vooren J. V. D. (2008). Introduction to sugarcane ethanol contributions to climate change mitigation and the environment. In: Zubbier P. & Vooren J. V. D. (Eds.). Sugarcane ethanol. Contributions to climate change mitigation and the environment. Wageningen: Wageningen Academic Publishers: 19-27.