In the context of rapidly advancing biotechnology, the Gibson Assembly technique is emerging as a breakthrough tool, enabling researchers to manipulate DNA easily without relying on traditional methods. Developed by Daniel G. Gibson and colleagues at the J. Craig Venter Institute (JCVI – California, USA) in 2009 (Gibson et al., 2009), this technique was published in the journal Nature Methods and quickly became a cornerstone in synthetic biology. With its ability to seamlessly assemble DNA in a single isothermal reaction, Gibson Assembly not only simplifies the process but also unlocks numerous potential applications, particularly in the field of food technology.
Gibson Assembly operates based on the coordination of three key enzymes: 5' exonuclease, DNA polymerase, and DNA ligase (Figure 1) (Gibson et al., 2009). The process occurs at a stable temperature of 50°C for 15-60 minutes. First, the exonuclease removes nucleotides from the 5' ends of DNA fragments, creating single-stranded overhangs at the overlapping regions (typically 20-40 bp). These overhangs then anneal to each other, the polymerase fills in the gaps, and the ligase seals the nicks, forming a seamless DNA molecule. This method outperforms traditional restriction enzyme techniques, which often leave sequence "scars" and require stringent conditions, being susceptible to DNA methylation or off-target activity.
    |
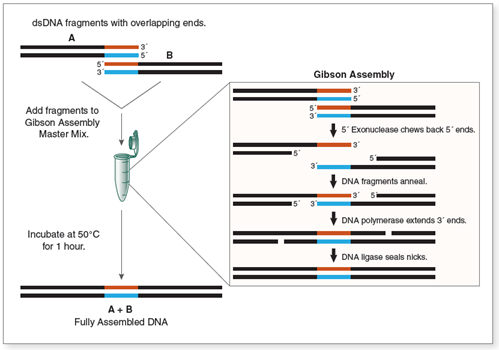 |
| Figure 1. Overview of the reactions occurring in Gibson Assembly |
A prominent application of Gibson Assembly is in cloning – the creation of recombinant DNA. The technique allows the assembly of multiple overlapping DNA fragments without the need for restriction enzyme sites, facilitating the flexible construction of expression vectors or plasmids (Gibson, 2011). For example, scientists at JCVI used this method to assemble the mouse mitochondrial genome (16.5 kb) from 600 60-base oligonucleotides (Gibson et al., 2010), or to synthesize the Mycoplasma mycoides genome (1.08 Mbp) – a milestone in creating the first synthetic cell (Gibson et al., 2010). In cloning, DNA can be divided into overlapping PCR fragments, then assembled into vectors such as pUC19 or pTYB1, and transformed into Escherichia coli for propagation. This is particularly useful for transferring genes from one organism to another, such as transferring biosynthetic pathways for compounds from bacteria to yeast (Figure 2).
    |
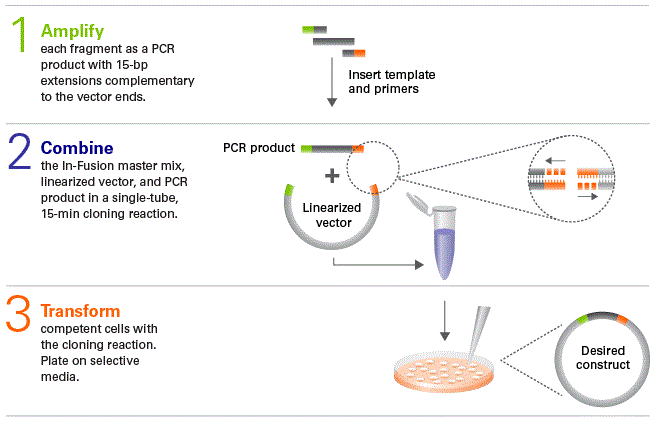 |
| Figure 2. Steps for gene transfer using the Gibson Assembly method |
Additionally, Gibson Assembly is widely applied in site-directed mutagenesis. By incorporating changes (substitutions, deletions, or insertions of nucleotides) into PCR primers at the overlapping regions, the technique enables rapid editing of large DNA molecules (Gibson, 2011). For instance, during the synthesis of the M. mycoides genome, 16 modular DNA fragments were edited to match the desired sequence (Gibson et al., 2010). With the capability to assemble multiple fragments simultaneously (up to 8-12) (Figure 3), the method can introduce 8 mutations across 80 kb of DNA in a single reaction. This facilitates gene optimization, such as enhancing protein expression or improving enzyme stability, without limitations imposed by DNA size (Gibson, 2011).
    |
 |
| Figure 3. Method for inserting 5 DNA fragments into a vector using Gibson Assembly |
In the field of food technology, Gibson Assembly is opening up promising application trends. The technique supports the development of next-generation probiotics by editing genes in lactic acid bacteria such as Lactiplantibacillus plantarum, enhancing disease resistance or the production of beneficial compounds (e.g., the anti-inflammatory protein Elafin) (Kumar et al., 2023). A recent study in the journal Vaccines (2023) utilized Gibson Assembly to create infectious clones of foot-and-mouth disease virus (FMDV) on the pKLS3 vector, supporting the production of customized vaccines for livestock – contributing to food security (Semkum et al., 2023). Furthermore, the method aids in assembling biosynthetic pathways for enzymes used in food (such as amylase and protease) or improving crop varieties resistant to pests and diseases, thereby increasing yields (New England Biolabs). In synthetic biology, it supports the creation of microorganisms that produce biofuels from agricultural byproducts or natural compounds like carotenoids and vitamins – aiming toward sustainable functional foods. Studies from NEB and Thermo Fisher emphasize that Gibson Assembly reduces experimental time, increases accuracy, and is suitable for large-scale production in the food industry (New England Biolabs).
However, the technique still has limitations, such as the requirement for precise primer design (Figure 4), reduced efficiency with short DNA fragments (<200 bp), or overlapping regions with secondary structures. To address these, improved versions like HiFi DNA Assembly have been developed, utilizing higher-fidelity polymerases (New England Biolabs).
    |
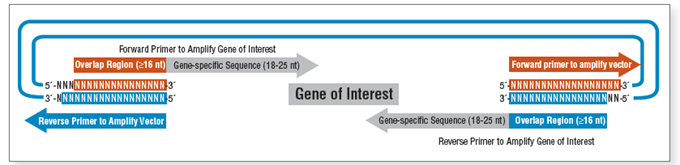 |
| Figure 4. Primer design requirements for the Gibson Assembly method |
In summary, Gibson Assembly not only revolutionizes biological research but also brings practical benefits to food technology, from improving food safety to sustainable production. With its flexible and efficient DNA assembly capabilities, this technique is opening up novel applications in synthetic biology, helping scientists design and edit genes more precisely. As a foundational tool, Gibson Assembly promises to continue driving scientific advancements, contributing to solving global challenges in the fields of food and biomedicine.
Dr. Le Thien-Kim
Faculty of Food Science and Technology
References
Gibson D. G. (2011). Enzymatic assembly of overlapping DNA fragments. Methods in Enzymology, 498, 349–361. DOI: 10.1016/B978-0-12-385120-8.00015-2.
Gibson D. G., Benders G. A., Andrews-Pfannkoch C., Denisova E. A., Baden-Tillson H., Zaveri J., Stockwell T. B., Brownley A., Thomas D. W., Algire M. A., Merryman C., Young L., Noskov V. N., Glass J. I., Venter J. C., Hutchison C. A. & Smith H. O. (2010). Creation of a bacterial cell controlled by a chemically synthesized genome. Science, 329(5987), 52–56. DOI: 10.1126/science.1190719.
Gibson D. G., Smith H. O., Hutchison C. A., Venter J. C. & Merryman C. (2010). Chemical synthesis of the mouse mitochondrial genome. Nature Methods, 7(11), 901–903. DOI: 10.1038/nmeth.1515.
Gibson D. G., Young L., Chuang R.-Y., Venter J. C., Hutchison C. A. & Smith H. O. (2009). Enzymatic assembly of DNA molecules up to several hundred kilobases. Nature Methods, 6(5), 343–345. DOI: 10.1038/nmeth.1318.
Kumar A., Andersson B., Barakat M. & Impens L. (2023). In vitro assembly of plasmid DNA for direct cloning in Lactiplantibacillus plantarum WCSF1. PLOS ONE, 18(2), e0281625. DOI: 10.1371/journal.pone.0281625.
New England Biolabs. (n.d.). Gibson Assembly® Cloning Kit. Retrieved from https://www.neb.com/en/products/e5510-gibson-assembly-cloning-kit.
New England Biolabs. (n.d.). NEBuilder® HiFi DNA Assembly. Retrieved from https://www.neb.com/en-us/applications/cloning-and-synthetic-biology/dna-assembly-and-cloning/nebuilder-hifi-dna-assembly.
Semkum P., Chungjatupornchai W., Sujjitjoon J., Tangjatturon N., Chatchaiphan S., Bunnueang N., Yamabhai M. & Eiamphungporn W. (2023). The application of the Gibson Assembly method in the production of two pKLS3 vector-derived infectious clones of foot-and-mouth disease virus. Vaccines, 11(6), 1111. DOI: 10.3390/vaccines11061111.